portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; visibility: visible; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; visibility: visible; overflow-wrap: break-word !important;">曆經近10年的“黃金”發展期,我國醫療器械國產(chan) 化率程度發展至何處?哪些設備實現從(cong) 0到1的突破,哪些賽道已經能夠與(yu) 外資巨頭並駕齊驅,又有哪些技術領域仍在“望其項背”奮力追趕中?或許從(cong) 下麵的一係列數據匯總,我們(men) 得以窺見醫療器械國產(chan) 替代的進程真相。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">從(cong) “0”到“1”,國產(chan) 醫械突破清單
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">截至 2024 年,在《醫療器械分類目錄》中的 1170 個(ge) 二級產(chan) 品類別下(注:不含體(ti) 外診斷試劑),產(chan) 品注冊(ce) 國產(chan) 化率(以國產(chan) 產(chan) 品注冊(ce) 國產(chan) 化率超過75%計)達67.01%,共有 784項二級產(chan) 品類別。而產(chan) 品注冊(ce) 國產(chan) 化率超過(含)50%的二級類別由 2020年的 872項增長至 2024年的 1011項。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">注:產(chan) 品注冊(ce) 國產(chan) 化率=國家藥監局分類目錄的二級類別下國產(chan) 產(chan) 品注冊(ce) 數/( 國產(chan) 產(chan) 品注冊(ce) 數+境外產(chan) 品注冊(ce) 數)*100%
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">表1 截至2024年國內(nei) 醫療器械二級產(chan) 品類別國產(chan) 化率區間分布
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; overflow-wrap: break-word !important;">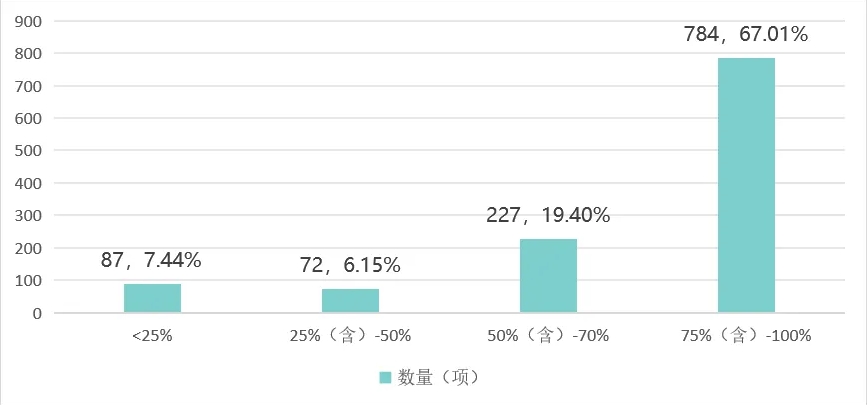
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">數據來源:高端醫械院數據中心、醫療器械創新網
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">表2 國產(chan) 注冊(ce) 占比≥50%的二級產(chan) 品類別數變化情況
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">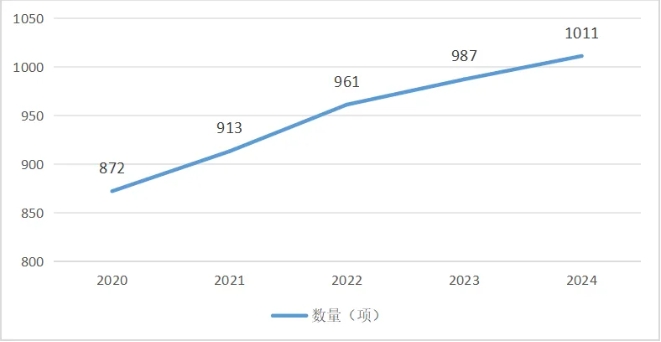
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">截至2024年底,在《醫療器械分類目錄》中的1170個(ge) 二級產(chan) 品類別下,我國有30項二級類別的產(chan) 品僅(jin) 有進口產(chan) 品注冊(ce) 證,尚未有國產(chan) 產(chan) 品獲批,這個(ge) 數字比2020年減少了37項,從(cong) 下麵數據不難看出我國產(chan) 品注冊(ce) 的國產(chan) 化進程從(cong) “0”到“1”的突破正穩紮穩打持續推進。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">表3 2020至2024年我國國產(chan) 注冊(ce) 證數量為(wei) 零的二級類別產(chan) 品數量
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">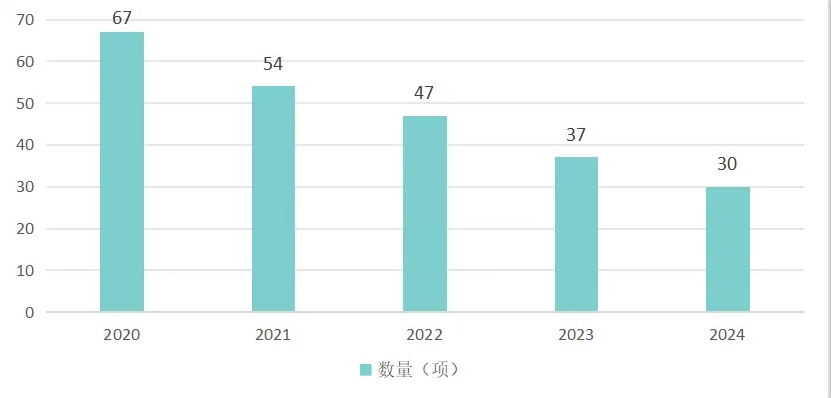
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">數據來源:高端醫械院數據中心、醫療器械創新網
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">從(cong) 具體(ti) 領域來看,我國共有6個(ge) 二級類別的產(chan) 品在2024年獲得了“零”的突破。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">表4 2024年國產(chan) 突破“零”的二級類別名單
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">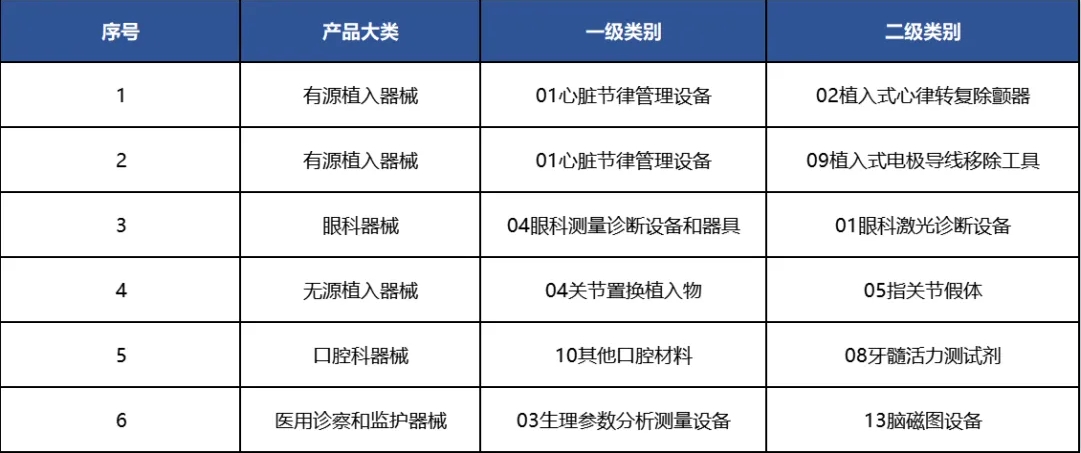
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">數據來源:高端醫械院數據中心、醫療器械創新網
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; overflow-wrap: break-word !important;">再細分來看,我國共有29個(ge) 二級類別的產(chan) 品在2024年僅(jin) 獲得1張國產(chan) 產(chan) 品注冊(ce) 證。
portant; overflow-wrap: break-word !important;">
portant; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">表5 2024年僅(jin) 獲得1張國產(chan) 產(chan) 品注冊(ce) 證二級類別名單
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">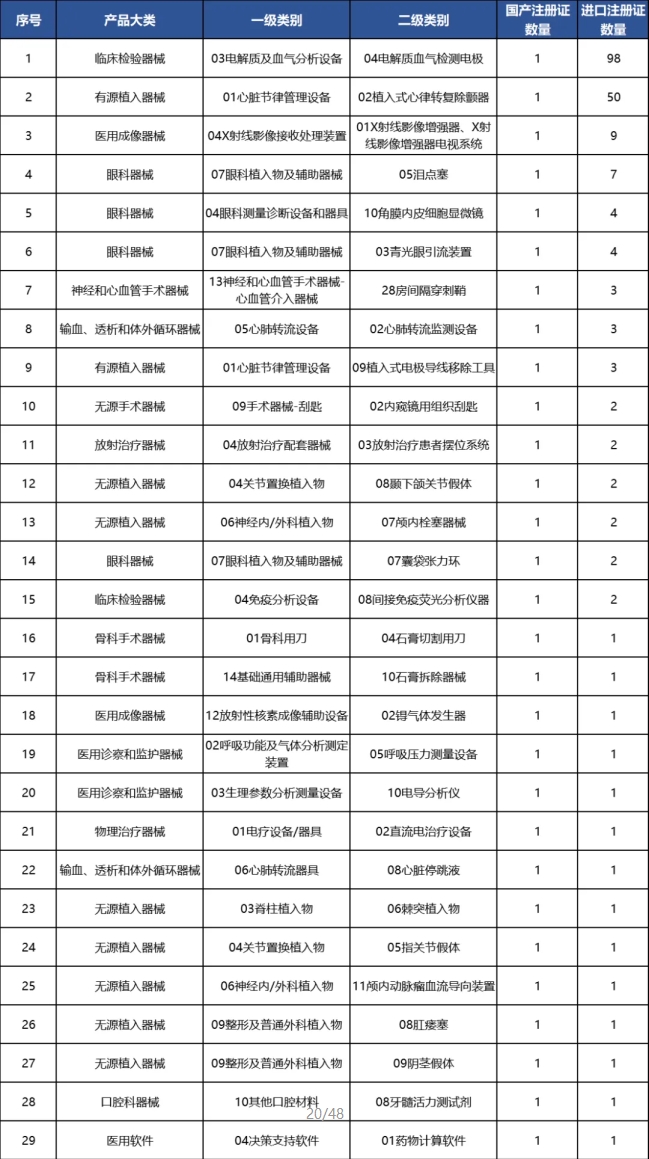
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">數據來源:高端醫械院數據中心、醫療器械創新網
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">尚未零突破的產(chan) 品主要集中在有源植入、放射治療和醫用成像領域,其中植入式心髒除顫電極導線最多,共有14款進口注冊(ce) 證獲批。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">表 6 截至2024年我國國產(chan) 注冊(ce) 證數量為(wei) 零的二級類別產(chan) 品名單
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; text-align: center; line-height: 1.75em; overflow-wrap: break-word !important;">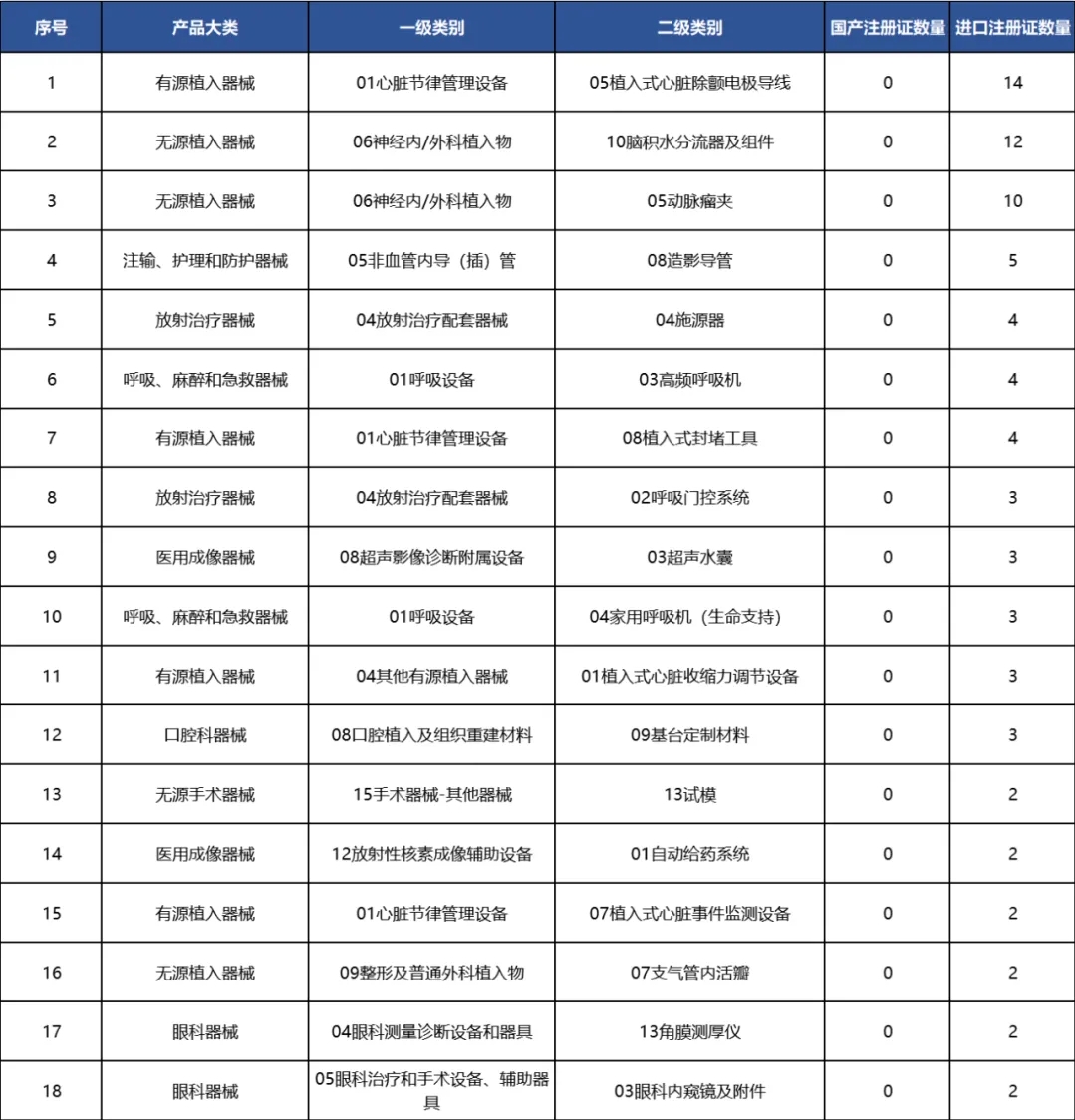
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">數據來源:高端醫械院數據中心、醫療器械創新網
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">從(cong) 10% 到 80% 的追趕之路充滿艱辛,而從(cong) 0 到 1% 的突破更為(wei) 艱難 —— 如同石縫中生長的幼苗,既要突破技術封鎖的岩層,又要適應市場檢驗的風雨,這也恰恰是國產(chan) 設備發展曆程的生動寫(xie) 照。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">成熟賽道,日漸由國產(chan) 主導的千億(yi) 市場
丨低值耗材:成熟期主導市場
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">
丨高值耗材:替代率超80%
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">
丨監護設備與生化診斷:中端市場突破
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">
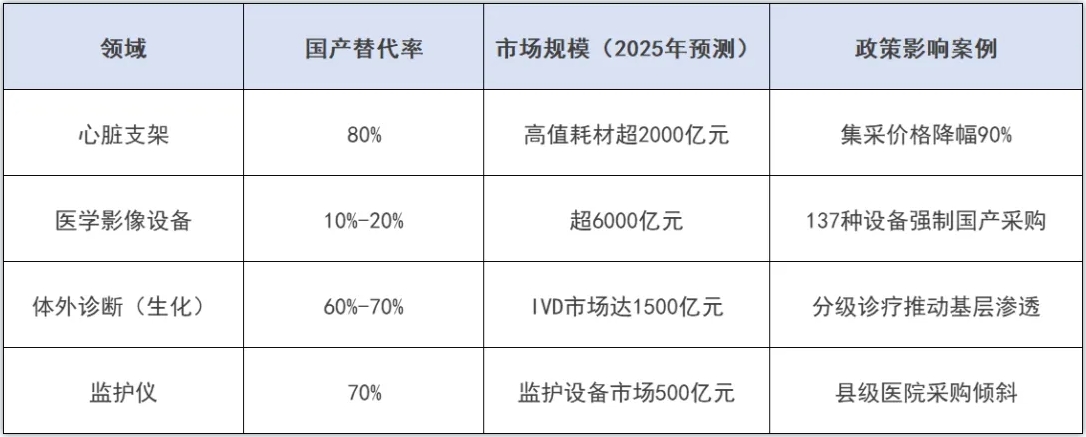
除了部分成熟度較高的產品國產化程度較高,還仍有大部分醫療器械設備市場占有率亟待提升,近年來隨著創新技術迭代與政策加持部分醫療設備已有突圍之勢。
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">
例如高端醫學影像設備,據統計當前,CT、MRI等高端醫學影像設備國產替代率僅10%-20%,外資品牌(如GE、西門子)仍壟斷三級醫院市場。但像聯影醫療等企業已實現技術突破,其PET-CT設備在國內新增市場占有率超30%,並逐步進入歐美市場。
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">政策驅動,國產(chan) 替代步入“攻堅期”
portant; border-radius: 0px; margin: 0pt 0pt 0pt 0px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">2023年,財政部與(yu) 工信部聯合發布的《政府采購進口產(chan) 品審核指導標準》對178種醫療器械設定國產(chan) 采購比例,其中137種要求100%國產(chan) 化,直接推動三級醫院采購轉向,2023年中央財政撥款200億(yi) 元支持縣級醫院設備采購,明確要求優(you) 先選擇國產(chan) 產(chan) 品。采購政策的傾(qing) 斜也是加速國產(chan) 替代的主要助力。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">此外,集采常態化也在加速國產(chan) 產(chan) 品崛起,其中心髒支架、人工關(guan) 節等品類通過集采價(jia) 格降幅超80%,倒逼外資退出中低端市場,國產(chan) 企業(ye) 憑借成本優(you) 勢占據主導。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">與(yu) 此同時,我國不斷加大對核心醫療器械零部件的技術攻克力度,其中科技部“十四五”重點研發計劃中,醫療設備專(zhuan) 項投入超50億(yi) 元,支持關(guan) 鍵零部件(如CT球管)攻關(guan) 。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">而醫療器械的快速發展也一定離不開審批流程的提質增效,近年來,創新醫療器械特別審批程序將產(chan) 品上市周期縮短30%,2024年共有62個(ge) 產(chan) 品通過該通道獲批,同比增加25%。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">走出去,探尋更全球廣闊天地,也成了國產(chan) 化發展的重要手段。目前我國正通過“一帶一路”推動國產(chan) 設備出口,2023年醫療設備出口額同比增長18%,東(dong) 南亞(ya) 成為(wei) 主要市場。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">當前,我國醫療器械國產(chan) 替代已從(cong) 低端耗材轉向高端設備,政策、技術與(yu) 資本形成合力,更讓人欣喜的是我們(men) 看到越來越多的產(chan) 品從(cong) 無到有,從(cong) 有到強,正逐步譜寫(xie) 屬於(yu) 自己的國產(chan) 品牌故事。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">未來5年,三級醫院的高端設備替代、產(chan) 業(ye) 鏈核心部件自主化以及全球化出海將成為(wei) 三大主線。企業(ye) 需聚焦臨(lin) 床需求差異化創新,政策端則需持續優(you) 化采購環境與(yu) 研發生態,在新機遇下搶占更多市場份額。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">後續我們(men) 還將從(cong) 市場占有率端詳細分析我國醫械設備國產(chan) 化進程,敬請期待。
portant; overflow-wrap: break-word !important;">
portant; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; letter-spacing: 0.578px; line-height: 1.75em; overflow-wrap: break-word !important;">高端醫械院數據中心,是國家四部委成果轉化示範基地設立的醫療器械產(chan) 業(ye) 大數據服務平台,基於(yu) 海量醫療器械行業(ye) 數據資源的集成與(yu) 治理,構建行業(ye) 數據基座,激活數據要素潛能,推動合規可信流通與(yu) 共享,為(wei) 各類創新主體(ti) 提供數據服務、數據產(chan) 品、數據谘詢等服務,降低行業(ye) 研發成本,為(wei) 深化醫療器械產(chan) 業(ye) 創新驅動、推動高質量發展提供有力支撐。
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; letter-spacing: 0.578px; line-height: 1.75em; overflow-wrap: break-word !important;">
portant; overflow-wrap: break-word !important;">
portant; border-radius: 0px; margin: 0px 8px; color: rgb(85, 85, 85); -webkit-tap-highlight-color: transparent; padding: 0px; outline: 0px; max-width: 100%; clear: both; min-height: 1em; line-height: 1.75em; overflow-wrap: break-word !important;">醫療器械創新網(www.innomd.org)是國家醫療器械產(chan) 業(ye) 技術創新聯盟匯同中國醫療器械行業(ye) 協會(hui) 創新服務專(zhuan) 委會(hui) 等若幹業(ye) 內(nei) 知名創新服務機構共同運營的專(zhuan) 業(ye) 化全創新鏈服務平台、也是中國醫療器械創新創業(ye) 大賽的共同主辦方。平台組織業(ye) 內(nei) 數百位專(zhuan) 家及各類創服機構,共同打造天津、蘇州、杭州、紹興(xing) 、西安等地醫療器械創新服務基地,集中展開40餘(yu) 項創新創業(ye) 服務。平台運營7年以來已服務5000多個(ge) 創新創業(ye) 項目,服務內(nei) 容包括各類創新創業(ye) 支持、產(chan) 品設計代工、投融資服務、品牌傳(chuan) 播、園區政府服務等創新鏈關(guan) 鍵環節,其服務受到多方讚譽,是國內(nei) 醫療器械創新創業(ye) 服務的頂級平台。
來源:醫療器械創新網